Study on the efficiency and mechanism of iron-modified cyanobacteria biochar in removing tetracycline from water
-
摘要:
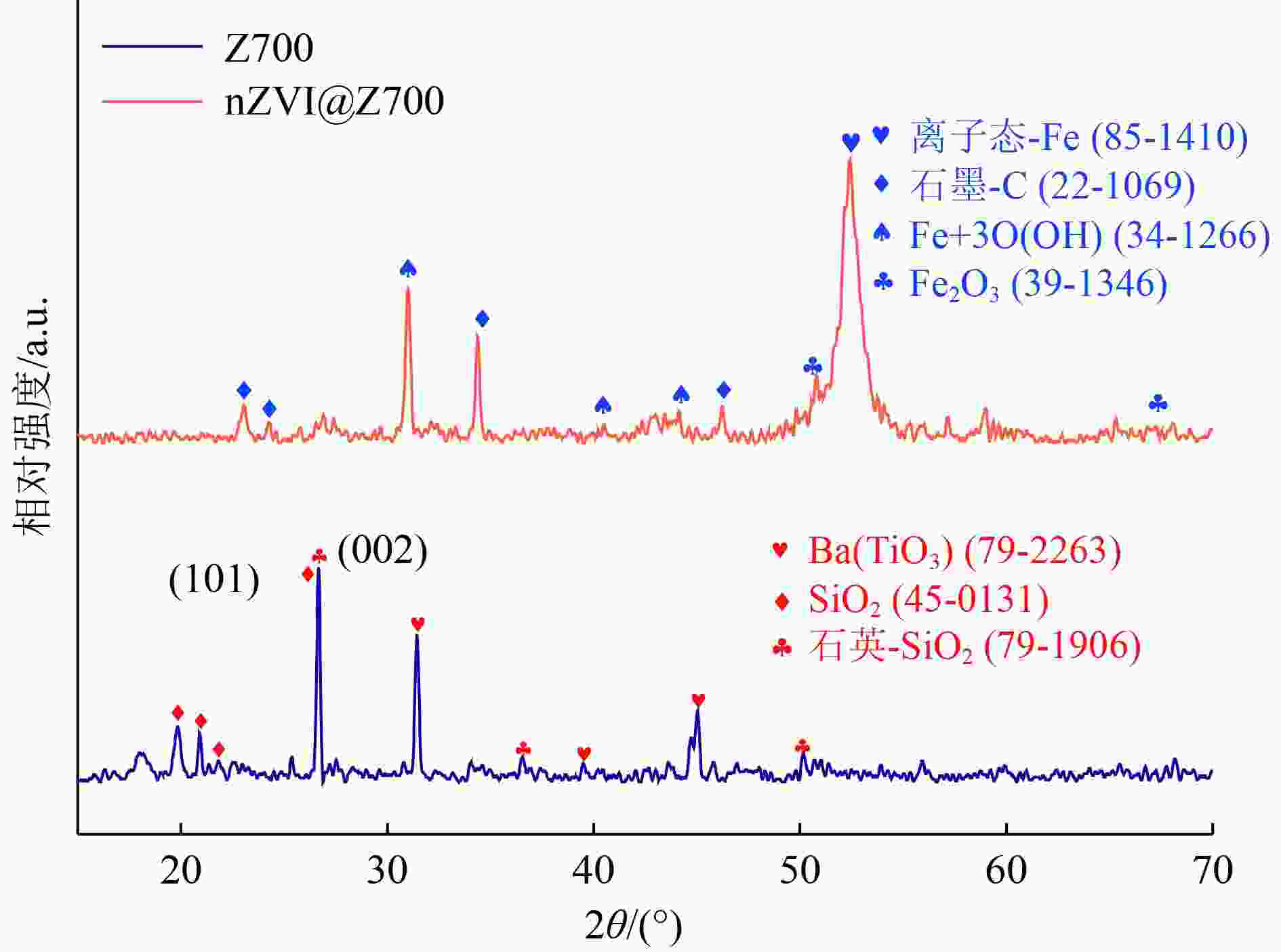
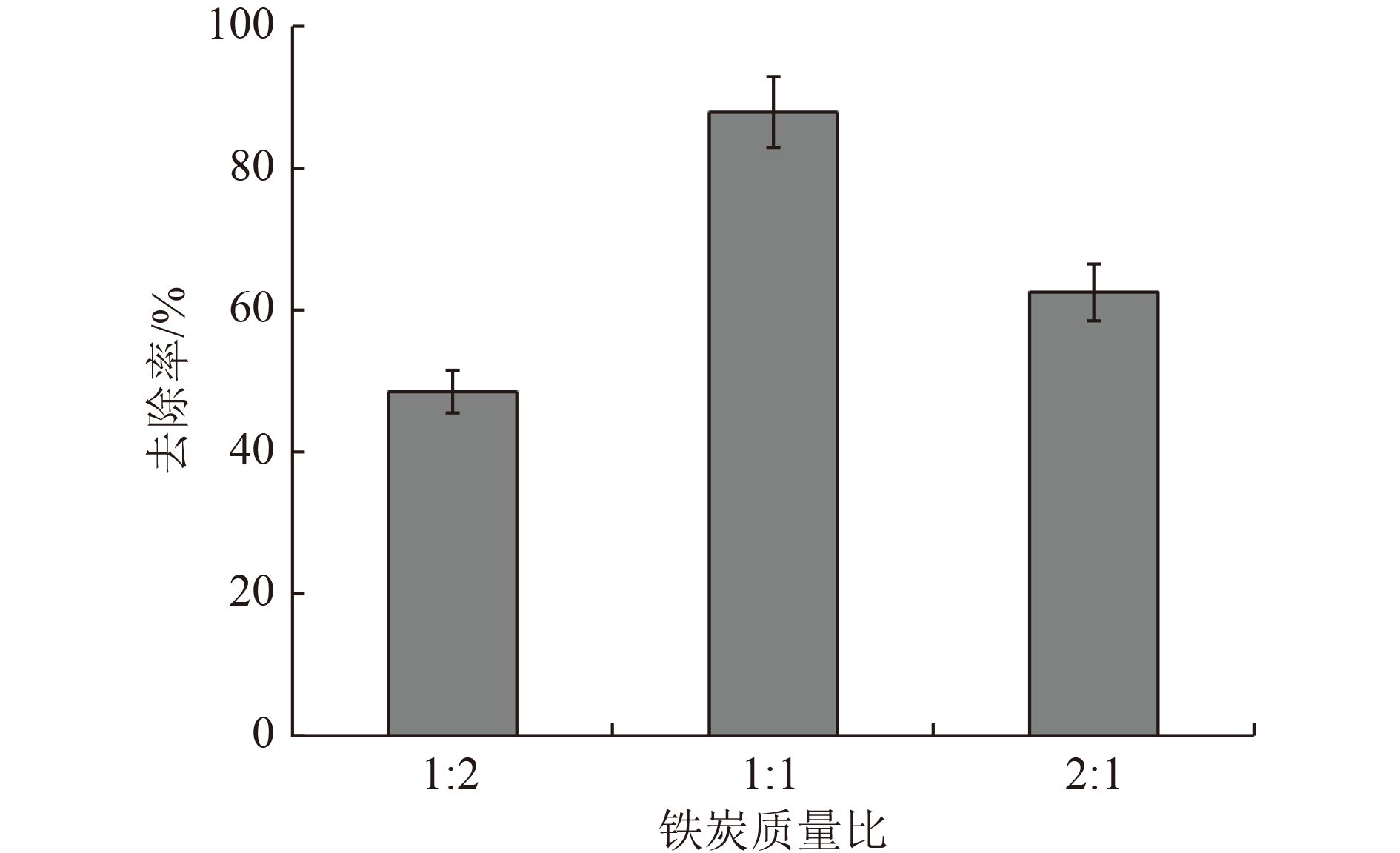
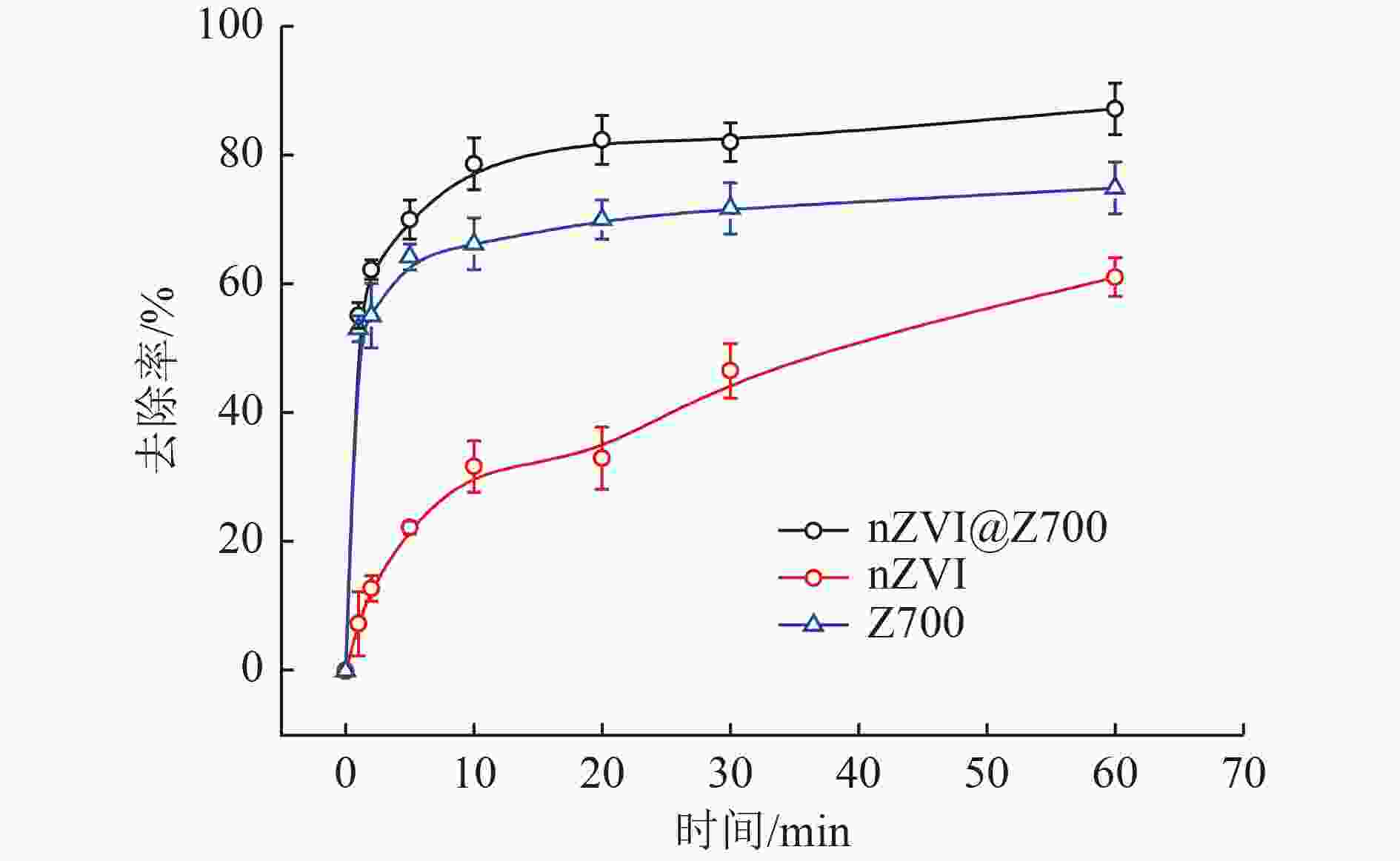
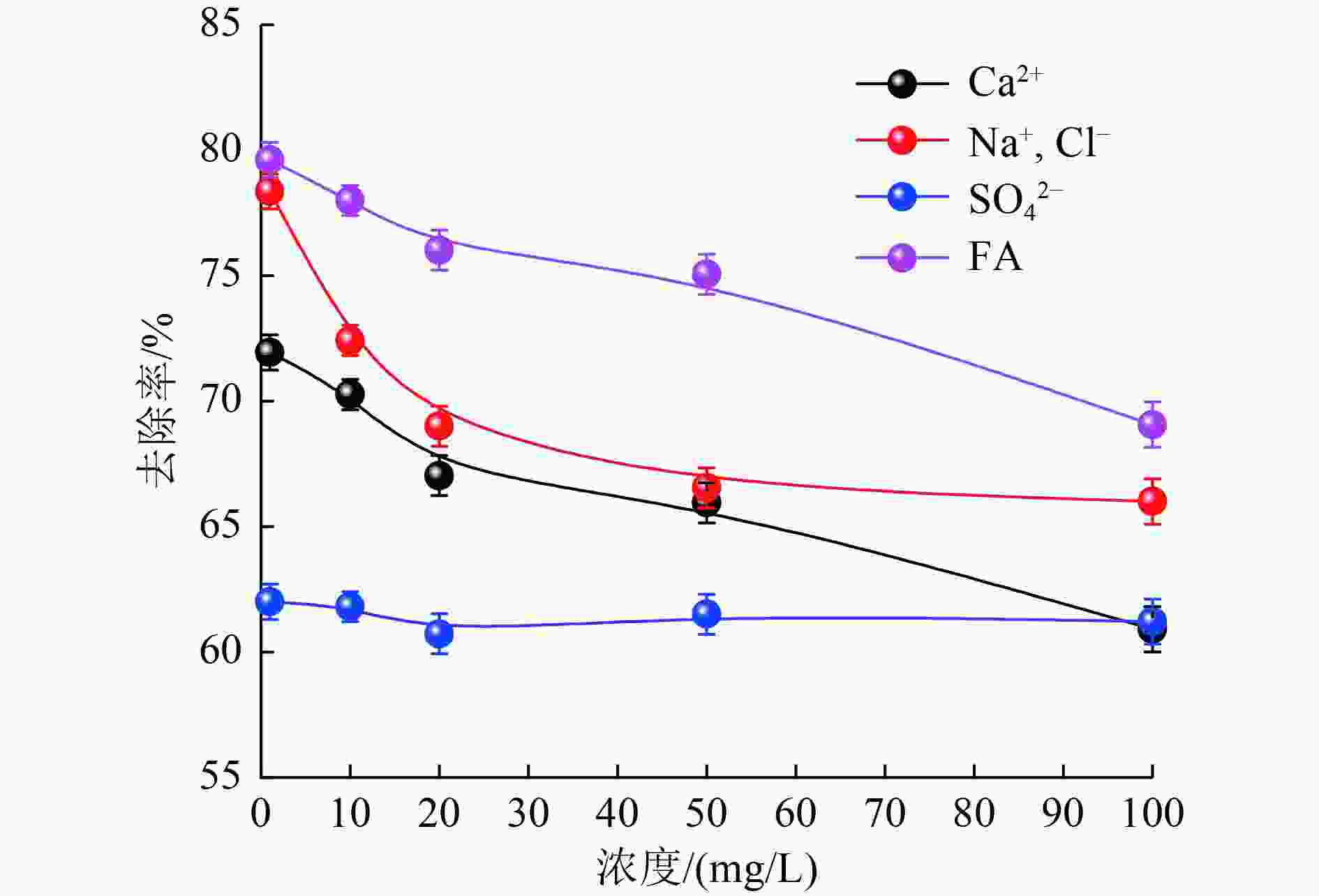
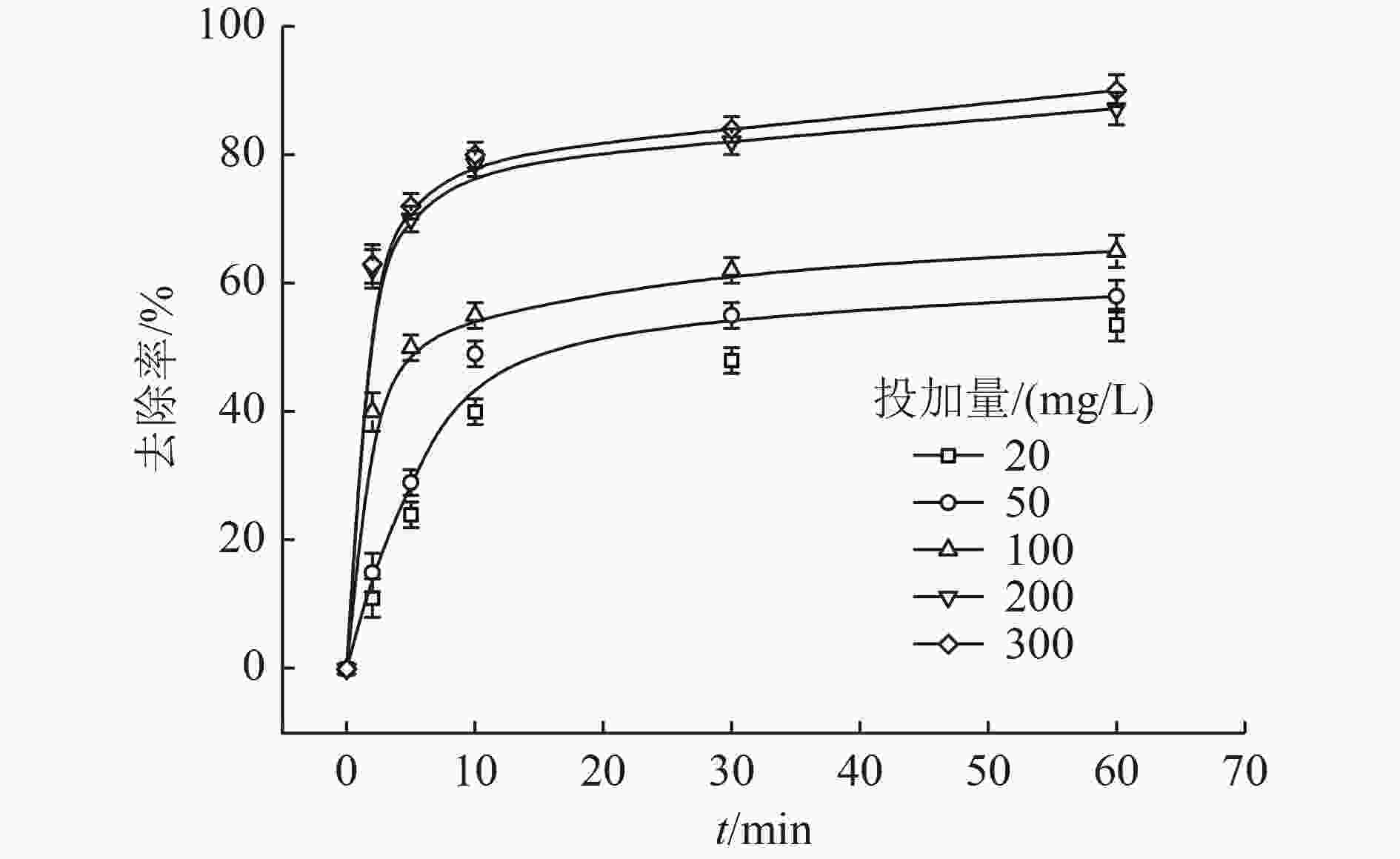
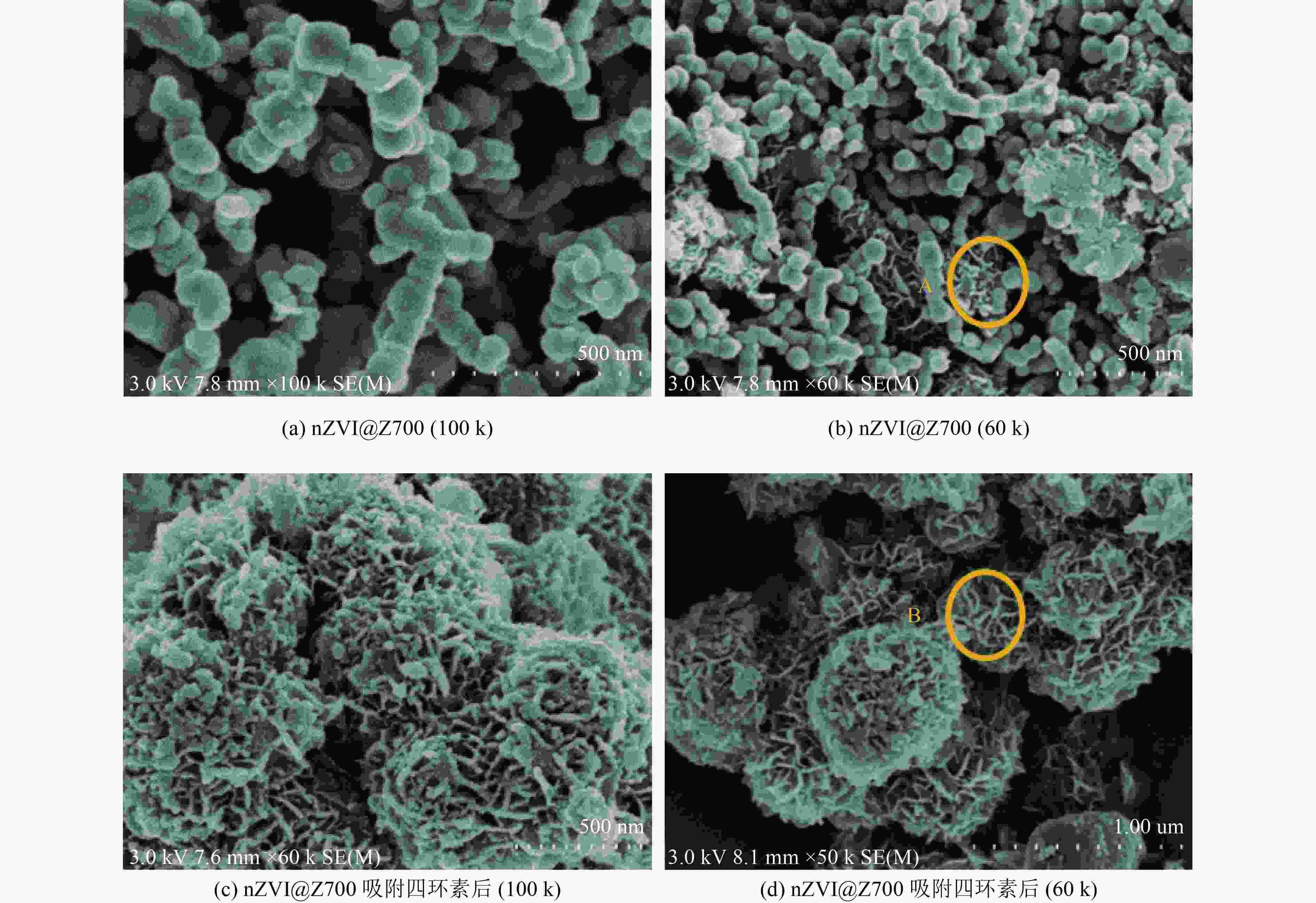
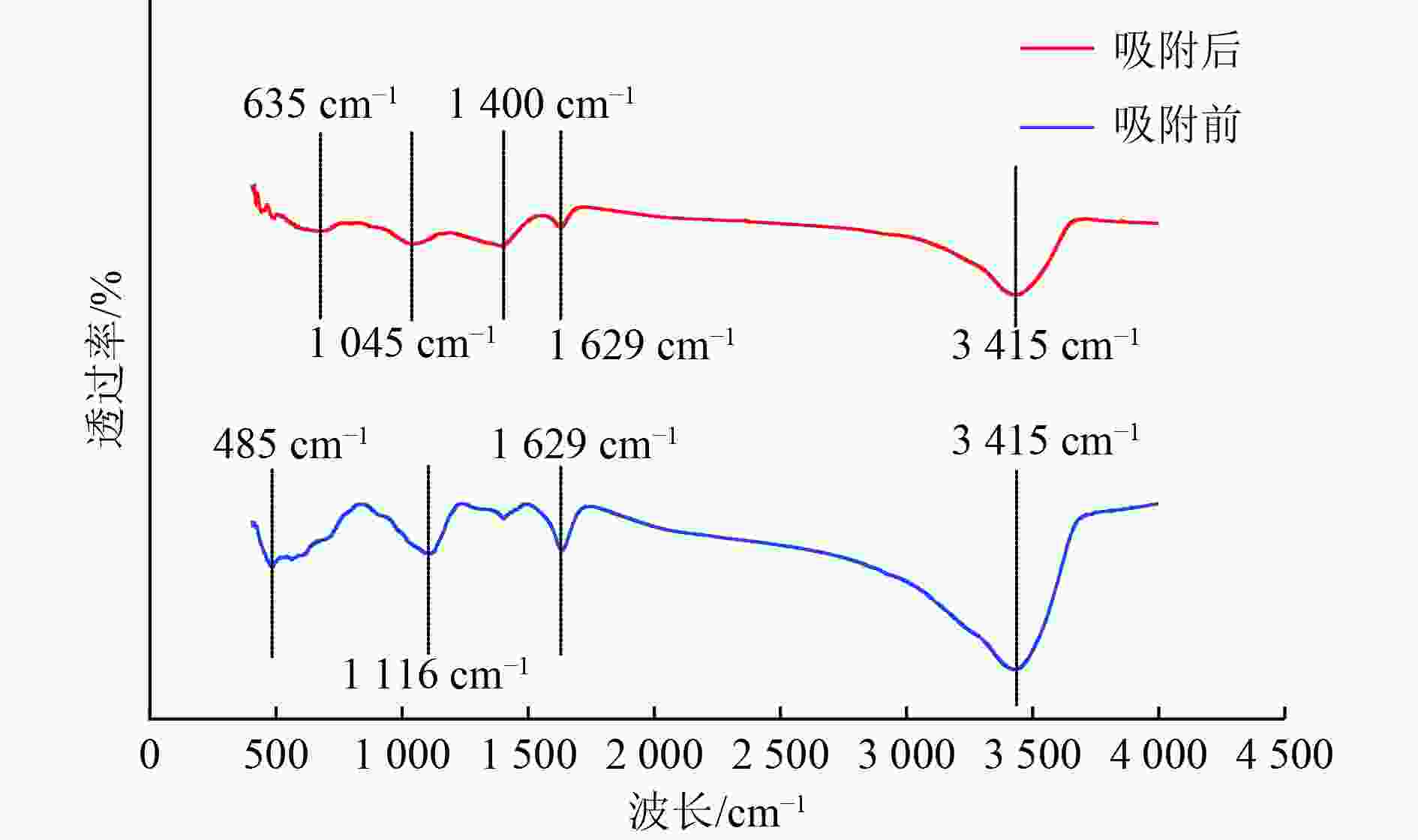
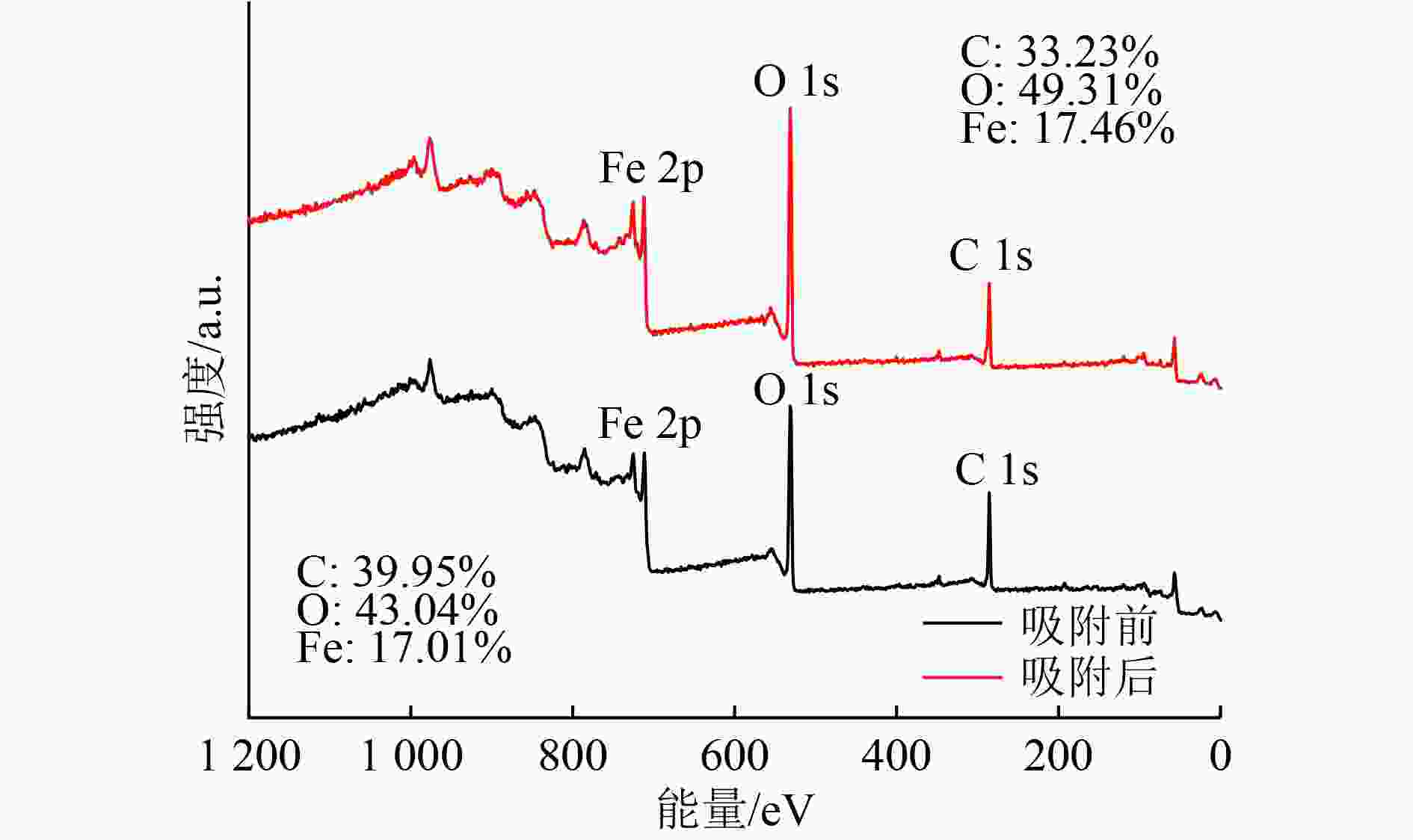
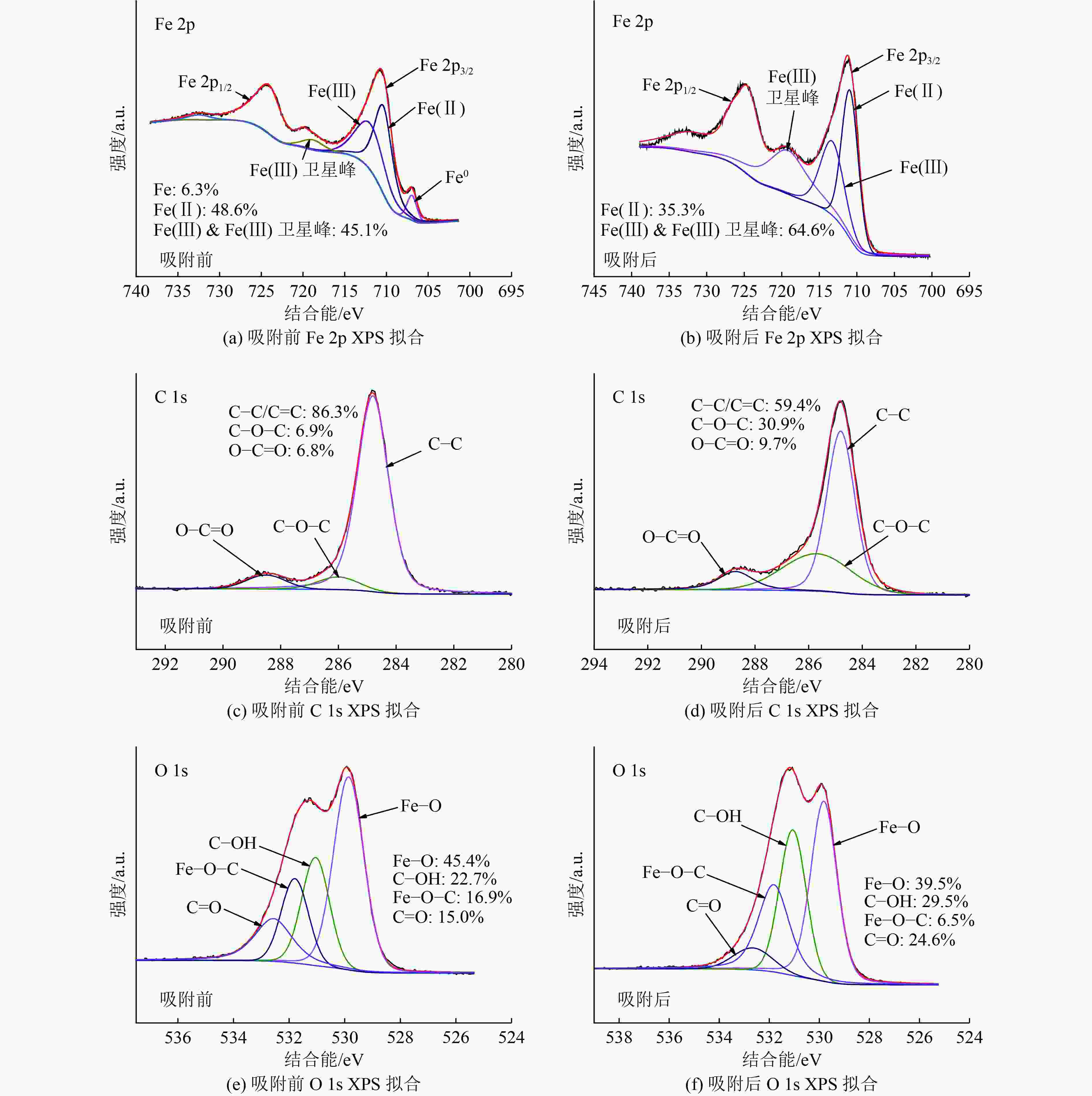
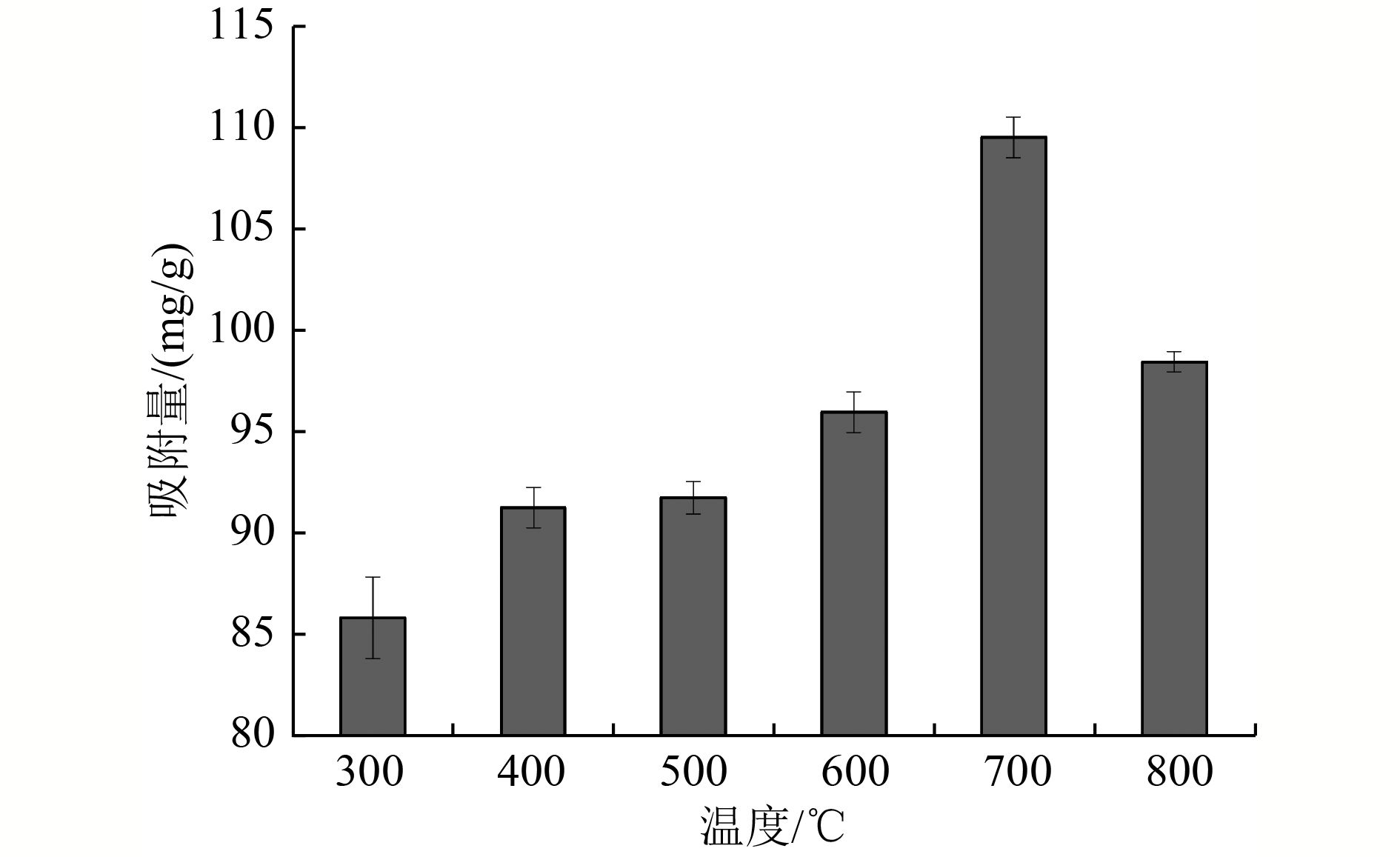
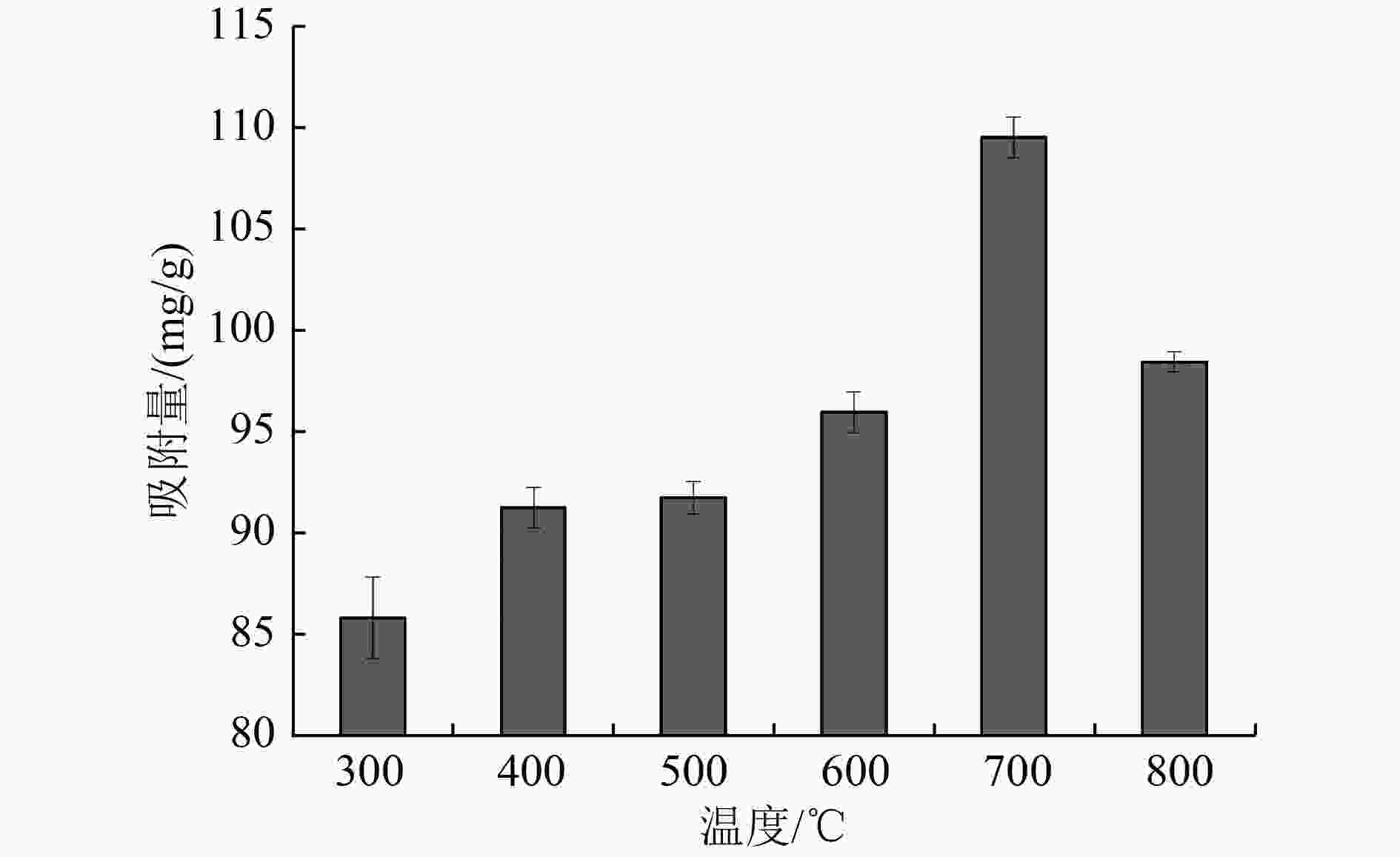
以蓝藻为原料制备生物炭,通过考察不同温度制备的蓝藻生物炭对四环素的吸附效能,筛选最优制备温度。采用液相还原法制备不同铁炭比的铁改性蓝藻生物炭,研究其对四环素的去除效能、影响因素及去除机理。结果表明:在700 ℃、铁炭质量比为1∶1条件下制备的铁改性蓝藻生物炭对四环素具有高效去除能力,60 min去除率可达87.2%,为改性前的1.2倍,吸附类型符合伪二级动力学方程(R2>0.99)。通过傅里叶红外光谱、扫描电镜、X射线光电子能谱、X射线衍射探讨铁改性蓝藻生物炭去除四环素性能与其结构的关系。结果表明,铁改性蓝藻生物炭对四环素的去除机理主要为吸附和化学降解作用,零价铁作为电子供体促进氧化还原反应的发生,含氧官能团作为电子转移桥梁在吸附降解过程中起着重要作用。影响因素试验结果表明,阴离子对铁改性蓝藻生物炭去除水中四环素效能的影响程度为SO4 2−>Cl−,阳离子影响程度为Ca2+>Na+,有机质黄腐酸相对于离子强度影响程度较弱。铁改性蓝藻生物炭对四环素类抗生素具有良好的去除能力,可为蓝藻资源化提供思路。
Abstract:Using cyanobacteria as raw material to prepare biochar, the adsorption efficiency of cyanobacteria biochar on tetracycline at different temperatures was investigated and the optimal preparation temperature of cyanobacteria biochar was screened. Cyanobacteria biochar modified with different Fe/C mass ratios was prepared by the liquid phase reduction method. The removing efficiency, influencing factors and removing mechanism of the iron-modified cyanobacteria biochar for tetracycline were studied. The results showed that the iron-modified cyanobacteria biochar had a high removing capacity for tetracycline at 700 ℃ and an iron-carbon mass ratio of 1∶1, and the removing rate reached 87.2% in 60 min, which was 1.2 times of that before modification. The adsorption type was in accordance with the pseudo-second-order kinetic equation (R2>0.99). The relationship between the iron-modified cyanobacteria biochar tetracycline removal performance and its structure was discussed by Fourier transform infrared spectroscopy (FTIR), scanning electron microscopy (SEM), X-ray photoelectron spectroscopy (XPS) and X-ray diffraction (XRD). It was proved that the main removal effects of tetracycline were adsorption and chemical degradation, the zero-valent iron as the electron donor promoted the redox reaction, and the oxygen-containing functional groups as the bridge of electron transfer played an important role in the adsorption and degradation process. The test results of influencing factors showed that the influence degree of anions was in the order of SO4 2− > Cl−, the influence degree of cations was Ca2+ > Na+, and the influence degree of organic fulvic acid (FA) was weak relative to ionic strength. The iron-modified cyanobacteria biochar had good removal efficiency on tetracycline antibiotics, which could provide a new idea for cyanobacteria resource utilization.
-
Key words:
- cyanobacteria /
- biochar /
- tetracycline /
- adsorption /
- degradation
-
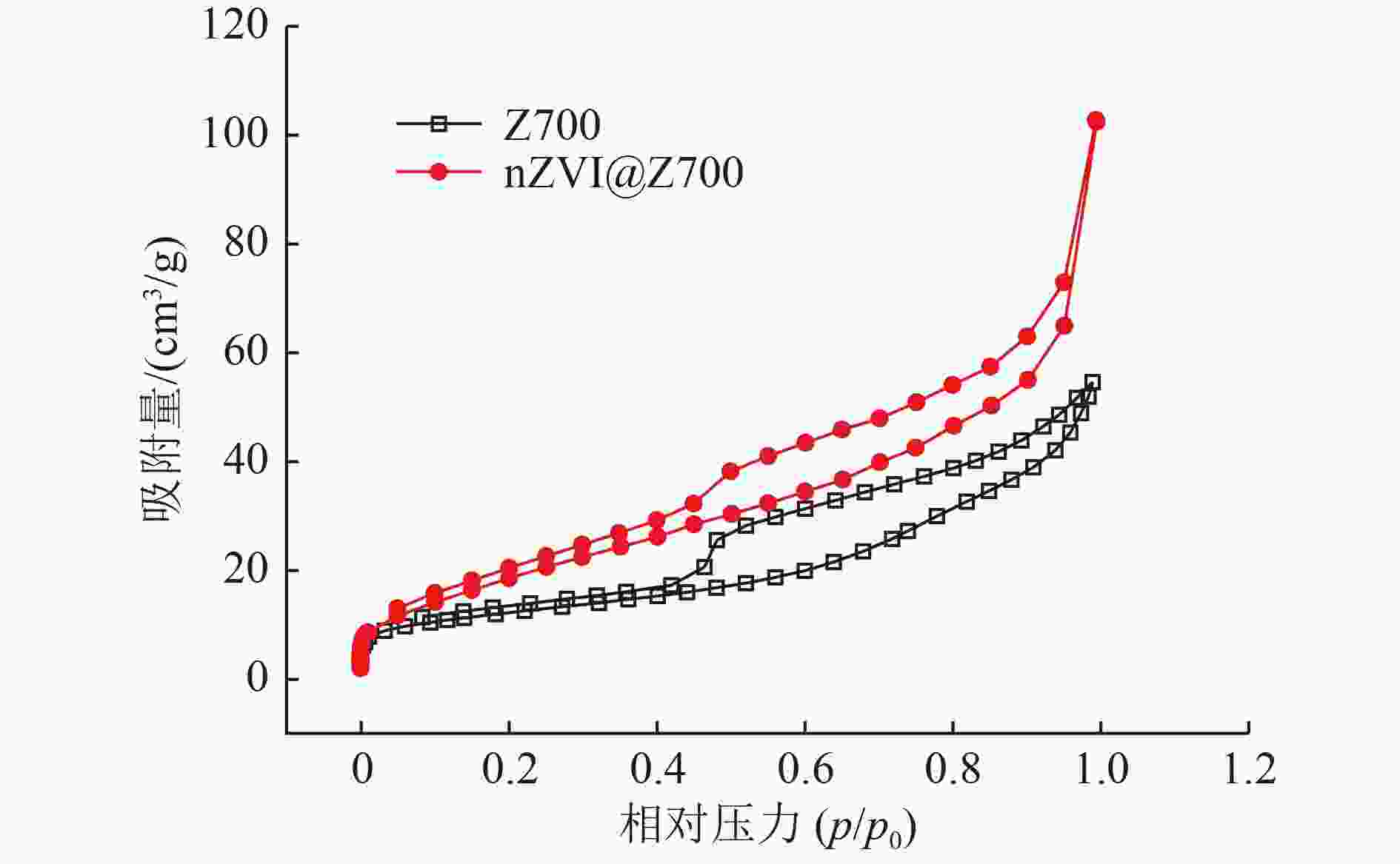
表 1 铁改性前后蓝藻生物炭的孔结构参数
Table 1. Pore structure parameters of cyanobacterial biochar before and after iron modification
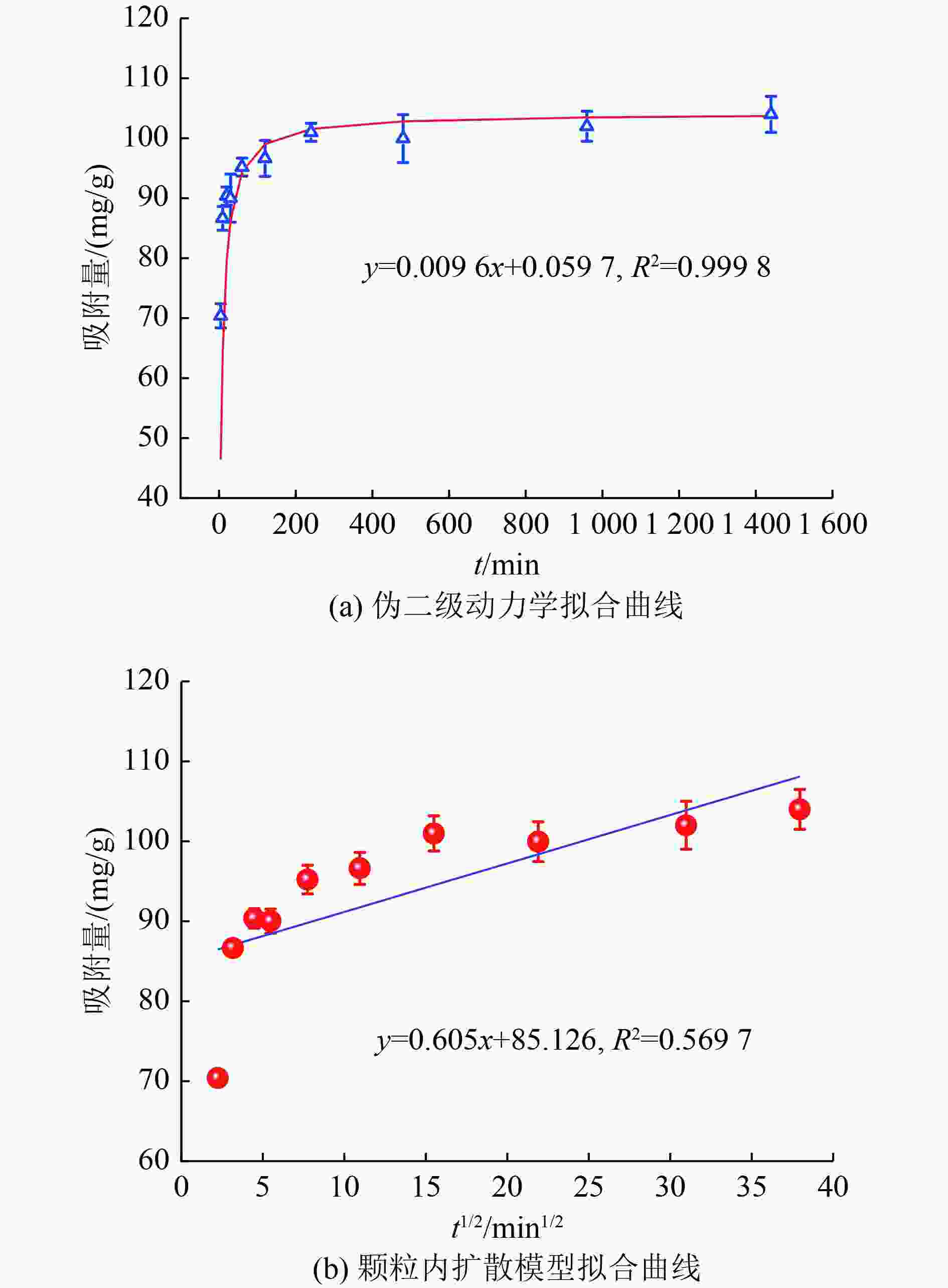
样品 孔径/nm 比表面积/(m2/g) 孔容积/(cm3/g) Z700 7.911 0 42.703 2 0.084 5 nZVI@Z700 8.977 7 70.592 0 0.158 4 表 2 nZVI@Z700去除四环素动力学参数
Table 2. Kinetic parameters of tetracycline removal by nZVI@Z700
伪一级动力学 伪二级动力学 颗粒内扩散模型 k1/
min−1R2 k2/
〔(mg/(g·min)〕qe
/(mg/g)R2 ki/
〔(mg/(g·min−1/2)〕R2 0.001 1×103 0.687 2 0.001 5×103 104.167 0 0.999 8 0.605 0 0.569 7 -
[1] TAN X F, LIU Y G, ZENG G M, et al. Application of biochar for the removal of pollutants from aqueous solutions[J]. Chemosphere,2015,125:70-85. doi: 10.1016/j.chemosphere.2014.12.058 [2] AHMAD M, RAJAPAKSHA A U, LIM J E, et al. Biochar as a sorbent for contaminant management in soil and water: a review[J]. Chemosphere,2014,99:19-33. doi: 10.1016/j.chemosphere.2013.10.071 [3] GOSWAMI R, SHIM J, DEKA S, et al. Characterization of cadmium removal from aqueous solution by biochar produced from Ipomoea fistulosa at different pyrolytic temperatures[J]. Ecological Engineering,2016,97:444-451. doi: 10.1016/j.ecoleng.2016.10.007 [4] LU H L, ZHANG W H, YANG Y X, et al. Relative distribution of Pb2+ sorption mechanisms by sludge-derived biochar[J]. Water Research,2012,46(3):854-862. doi: 10.1016/j.watres.2011.11.058 [5] CANTRELL K B, HUNT P G, UCHIMIYA M, et al. Impact of pyrolysis temperature and manure source on physicochemical characteristics of biochar[J]. Bioresource Technology,2012,107:419-428. doi: 10.1016/j.biortech.2011.11.084 [6] QI F J, YAN Y B, LAMB D, et al. Thermal stability of biochar and its effects on cadmium sorption capacity[J]. Bioresource Technology,2017,246:48-56. doi: 10.1016/j.biortech.2017.07.033 [7] 谢伟雪, 刘孝敏, 李小东, 等.废毛发生物炭的特性及其对Ni(Ⅱ)和Zn(Ⅱ)的吸附研究[J]. 环境工程技术学报,2018,8(6):656-661. doi: 10.3969/j.issn.1674-991X.2018.06.087XIE W X, LIU X M, LI X D, et al. Characteristics of waste hair biochar and its adsorption to Ni(Ⅱ) and Zn(Ⅱ)[J]. Journal of Environmental Engineering Technology,2018,8(6):656-661. doi: 10.3969/j.issn.1674-991X.2018.06.087 [8] 罗智宇, 赵野, 胡利华, 等.太湖蓝藻治理策略探讨[J]. 环境生态学,2019,1(4):45-48.LUO Z Y, ZHAO Y, HU L H, et al. Discussion on algae management strategy in Taihu lake[J]. Environmental Ecology,2019,1(4):45-48. [9] 殷鹏, 张建华, 孔繁璠.太湖蓝藻无害化处置资源化利用现状分析与对策研究[J]. 江苏水利,2019(9):23-25.YIN P, ZHANG J H, KONG F F. Current situation analysis and countermeasure research on the harmless disposal and resource utilization of cyanobacteria in Taihu Lake[J]. Jiangsu Water Resources,2019(9):23-25. [10] YU K L, LAU B F, SHOW P L, et al. Recent developments on algal biochar production and characterization[J]. Bioresource Technology,2017,246:2-11. doi: 10.1016/j.biortech.2017.08.009 [11] HUNG C M, HUANG C P, HSIEH S L, et al. Biochar derived from red algae for efficient remediation of 4-nonylphenol from marine sediments[J]. Chemosphere,2020,254:126916. doi: 10.1016/j.chemosphere.2020.126916 [12] AHMED M J, OKOYE P U, HUMMADI E H, et al. High-performance porous biochar from the pyrolysis of natural and renewable seaweed (Gelidiella acerosa) and its application for the adsorption of methylene blue[J]. Bioresource Technology,2019,278:159-164. doi: 10.1016/j.biortech.2019.01.054 [13] FAZAL T, RAZZAQ A, JAVED F, et al. Integrating adsorption and photocatalysis: a cost effective strategy for textile wastewater treatment using hybrid biochar-TiO2 composite[J]. Journal of Hazardous Materials,2020,390:121623. doi: 10.1016/j.jhazmat.2019.121623 [14] YAO X X, JI L L, GUO J, et al. Magnetic activated biochar nanocomposites derived from wakame and its application in methylene blue adsorption[J]. Bioresource Technology,2020,302:122842. doi: 10.1016/j.biortech.2020.122842 [15] 史宸菲, 李雨濛, 冯瑞杰, 等.蓝藻生物炭的制备及对过硫酸盐的活化效能[J]. 生态与农村环境学报,2017,33(12):1140-1145. doi: 10.11934/j.issn.1673-4831.2017.12.011SHI C F, LI Y M, FENG R J, et al. Preparation of biochar from cyanobacteria and function of the biochar for persulfate activation[J]. Journal of Ecology and Rural Environment,2017,33(12):1140-1145. doi: 10.11934/j.issn.1673-4831.2017.12.011 [16] ZHAO Y X, CHI Y T, TIAN C, et al. Recycling of titanium-coagulated algae-rich sludge for enhanced photocatalytic oxidation of phenolic contaminants through oxygen vacancy[J]. Water Research,2020,177:115789. doi: 10.1016/j.watres.2020.115789 [17] LIU P Y, RAO D A, ZOU L Y, et al. Capacity and potential mechanisms of Cd(Ⅱ) adsorption from aqueous solution by blue algae-derived biochars[J]. Science of the Total Environment,2021,767:145447. doi: 10.1016/j.scitotenv.2021.145447 [18] ZENG S Q, KAN E. Chemical activation of forage grass-derived biochar for treatment of aqueous antibiotic sulfamethoxazole[J]. American Chemical Society Omega,2020,5(23):13793-13801. [19] LI A Y, DENG H, JIANG Y H, et al. Superefficient removal of heavy metals from wastewater by Mg-loaded biochars: adsorption characteristics and removal mechanisms[J]. Langmuir,2020,36(31):9160-9174. doi: 10.1021/acs.langmuir.0c01454 [20] PEI X Y, PENG X X, JIA X S, et al. N-doped biochar from sewage sludge for catalytic peroxydisulfate activation toward sulfadiazine: efficiency, mechanism, and stability[J]. Journal of Hazardous Materials,2021,419:126446. doi: 10.1016/j.jhazmat.2021.126446 [21] 李华夏, 林毅, 周小斌, 等. 生物炭负载纳米零价铁去除废水中重金属的研究进展[J]. 环境工程技术学报, 2022, 12(3): 787-793.
LI H X, LIN Y, ZHOU X B, et al. Research progress on heavy metals removal from wastewater by biochar-supported nano zero-valent iron[J]. Journal of Environmental Engineering Technology, 2022, 12(3): 787-793.[22] HUO Y, LI W, MIN D, et al. Zero-valent iron nanoparticles with sustained high reductive activity for carbon tetrachloride dechlorination[J]. Royal Society of Chemistry Advances,2015,5(67):54497-54504. [23] 庞新宇, 刘文士, 李猛, 等.生物炭环境修复应用研究的文献计量学分析[J]. 环境工程技术学报,2021,11(4):740-749. doi: 10.12153/j.issn.1674-991X.20200261PANG X Y, LIU W S, LI M, et al. Research progress of biochar's application in environmental remediation based on bibliometrics[J]. Journal of Environmental Engineering Technology,2021,11(4):740-749. doi: 10.12153/j.issn.1674-991X.20200261 [24] YING B, LIN G L, JIN L S, et al. Adsorption and degradation of 2, 4-dichlorophenoxyacetic acid in spiked soil with Fe0 nanoparticles supported by biochar[J]. Acta Agriculturae Scandinavica, Section B:Soil & Plant Science,2015,65(3):215-221. [25] HWANG Y H, KIM D G, SHIN H S. Effects of synthesis conditions on the characteristics and reactivity of nano scale zero valent iron[J]. Applied Catalysis B:Environmental,2011,105(1/2):144-150. [26] 冯丽, 葛小鹏, 王东升, 等.pH值对纳米零价铁吸附降解2, 4-二氯苯酚的影响[J]. 环境科学,2012,33(1):94-103.FENG L, GE X P, WANG D S, et al. Effects of pH value on the adsorption and degradation of 2, 4-DCP by nanoscale zero-valent iron[J]. Environmental Science,2012,33(1):94-103. [27] WEI J, LIU Y T, LI J, et al. Adsorption and co-adsorption of tetracycline and doxycycline by one-step synthesized iron loaded sludge biochar[J]. Chemosphere,2019,236:124254. doi: 10.1016/j.chemosphere.2019.06.224 [28] CHEN J H, YU X L, LI C, et al. Removal of tetracycline via the synergistic effect of biochar adsorption and enhanced activation of persulfate[J]. Chemical Engineering Journal,2020,382:122916. doi: 10.1016/j.cej.2019.122916 [29] CHENG D L, NGO H H, GUO W S, et al. Feasibility study on a new pomelo peel derived biochar for tetracycline antibiotics removal in swine wastewater[J]. Science of the Total Environment,2020,720:137662. doi: 10.1016/j.scitotenv.2020.137662 [30] LI H Q, HU J T, MENG Y, et al. An investigation into the rapid removal of tetracycline using multilayered graphene-phase biochar derived from waste chicken feather[J]. Science of the Total Environment,2017,603/604:39-48. doi: 10.1016/j.scitotenv.2017.06.006 [31] SHEN L, ZHANG L H, WANG K, et al. Analysis of oxidation degree of graphite oxide and chemical structure of corresponding reduced graphite oxide by selecting different-sized original graphite[J]. Royal Society of Chemistry Advances,2018,8(31):17209-17217. [32] LI Y, GAO L M, LU Z X, et al. Enhanced removal of heavy metals from water by Hydrous ferric oxide-modified biochar[J]. American Chemical Society Omega,2020,5(44):28702-28711. [33] DONG H R, DENG J M, XIE Y K, et al. Stabilization of nanoscale zero-valent iron (nZVI) with modified biochar for Cr(Ⅵ) removal from aqueous solution[J]. Journal of Hazardous Materials,2017,332:79-86. doi: 10.1016/j.jhazmat.2017.03.002 [34] PI Z J, LI X M, WANG D B, et al. Persulfate activation by oxidation biochar supported magnetite particles for tetracycline removal: performance and degradation pathway[J]. Journal of Cleaner Production,2019,235:1103-1115. doi: 10.1016/j.jclepro.2019.07.037 [35] 俞花美. 生物质炭对环境中阿特拉津的吸附解吸作用及机理研究[D]. 北京: 中国矿业大学(北京), 2014. [36] WANG T, XUE L, ZHENG L W, et al. Biomass-derived N/S dual-doped hierarchically porous carbon material as effective adsorbent for the removal of bisphenol F and bisphenol S[J]. Journal of Hazardous Materials,2021,416:126126. doi: 10.1016/j.jhazmat.2021.126126 [37] 吴志坚, 刘海宁, 张慧芳.离子强度对吸附影响机理的研究进展[J]. 环境化学,2010,29(6):997-1003.WU Z J, LIU H N, ZHANG H F. Research progress on mechanisms about the effect of ionic strength on adsorption[J]. Environmental Chemistry,2010,29(6):997-1003. [38] LUO H Y, LIU Y, LU H X, et al. Efficient adsorption of tetracycline from aqueous solutions by modified alginate beads after the removal of Cu(Ⅱ) ions[J]. American Chemical Society Omega,2021,6(9):6240-6251. [39] PEIRIS C, GUNATILAKE S R, MLSNA T E, et al. Biochar based removal of antibiotic sulfonamides and tetracyclines in aquatic environments: a critical review[J]. Bioresource Technology,2017,246:150-159. doi: 10.1016/j.biortech.2017.07.150 [40] DAI J W, MENG X F, ZHANG Y Z, et al. Effects of modification and magnetization of rice straw derived biochar on adsorption of tetracycline from water[J]. Bioresource Technology,2020,311:123455. doi: 10.1016/j.biortech.2020.123455 [41] LÜTZENKIRCHEN J. Ionic strength effects on cation sorption to oxides: macroscopic observations and their significance in microscopic interpretation[J]. Journal of Colloid and Interface Science,1997,195(1):149-155. doi: 10.1006/jcis.1997.5160 [42] WU Y W, YUE Q Y, REN Z F, et al. Immobilization of nanoscale zero-valent iron particles (nZVI) with synthesized activated carbon for the adsorption and degradation of Chloramphenicol (CAP)[J]. Journal of Molecular Liquids,2018,262:19-28. doi: 10.1016/j.molliq.2018.04.032 [43] WANG H Z, GUO W Q, LIU B H, et al. Sludge-derived biochar as efficient persulfate activators: sulfurization-induced electronic structure modulation and disparate nonradical mechanisms[J]. Applied Catalysis B:Environmental,2020,279:119361. doi: 10.1016/j.apcatb.2020.119361 [44] 陈华, 任晓惠, 罗汉金, 等.改性纳米零价铁的制备及其去除水中的四环素[J]. 环境工程学报,2011,5(4):767-771.CHEN H, REN X H, LUO H J, et al. Preparation of modified nanoscale zero-valent iron particles and its application in removal of tetracycline from wastewater[J]. Chinese Journal of Environmental Engineering,2011,5(4):767-771. [45] GONG C, CHEN F, YANG Q, et al. Heterogeneous activation of peroxymonosulfate by Fe-Co layered doubled hydroxide for efficient catalytic degradation of Rhoadmine B[J]. Chemical Engineering Journal,2017,321:222-232. doi: 10.1016/j.cej.2017.03.117 [46] CHEN F, YANG Q, WANG S N, et al. Graphene oxide and carbon nitride nanosheets co-modified silver chromate nanoparticles with enhanced visible-light photoactivity and anti-photocorrosion properties towards multiple refractory pollutants degradation[J]. Applied Catalysis B:Environmental,2017,209:493-505. doi: 10.1016/j.apcatb.2017.03.026 [47] 朱佳燕, 张漓杉, 钟山, 等.粒状纳米零价铁/炭的制备及对孔雀绿的降解机理[J]. 环境化学,2021,40(5):1514-1523. doi: 10.7524/j.issn.0254-6108.2020102103ZHU J Y, ZHANG L Z, ZHONG S, et al. Removal of malachite green from wastewater by zero-valent iron nanoparticles supported on activated carbon powder[J]. Environmental Chemistry,2021,40(5):1514-1523. doi: 10.7524/j.issn.0254-6108.2020102103 [48] LAI B, ZHOU Y, YANG P, et al. Degradation of 3, 3'-iminobis-propanenitrile in aqueous solution by Fe(0)/GAC micro-electrolysis system[J]. Chemosphere,2013,90(4):1407-1477. ⊗ -




 下载:
下载: