Preparation of Hangjin clay-supported sulfidated zero-valent iron and its performance on phosphate removal
-
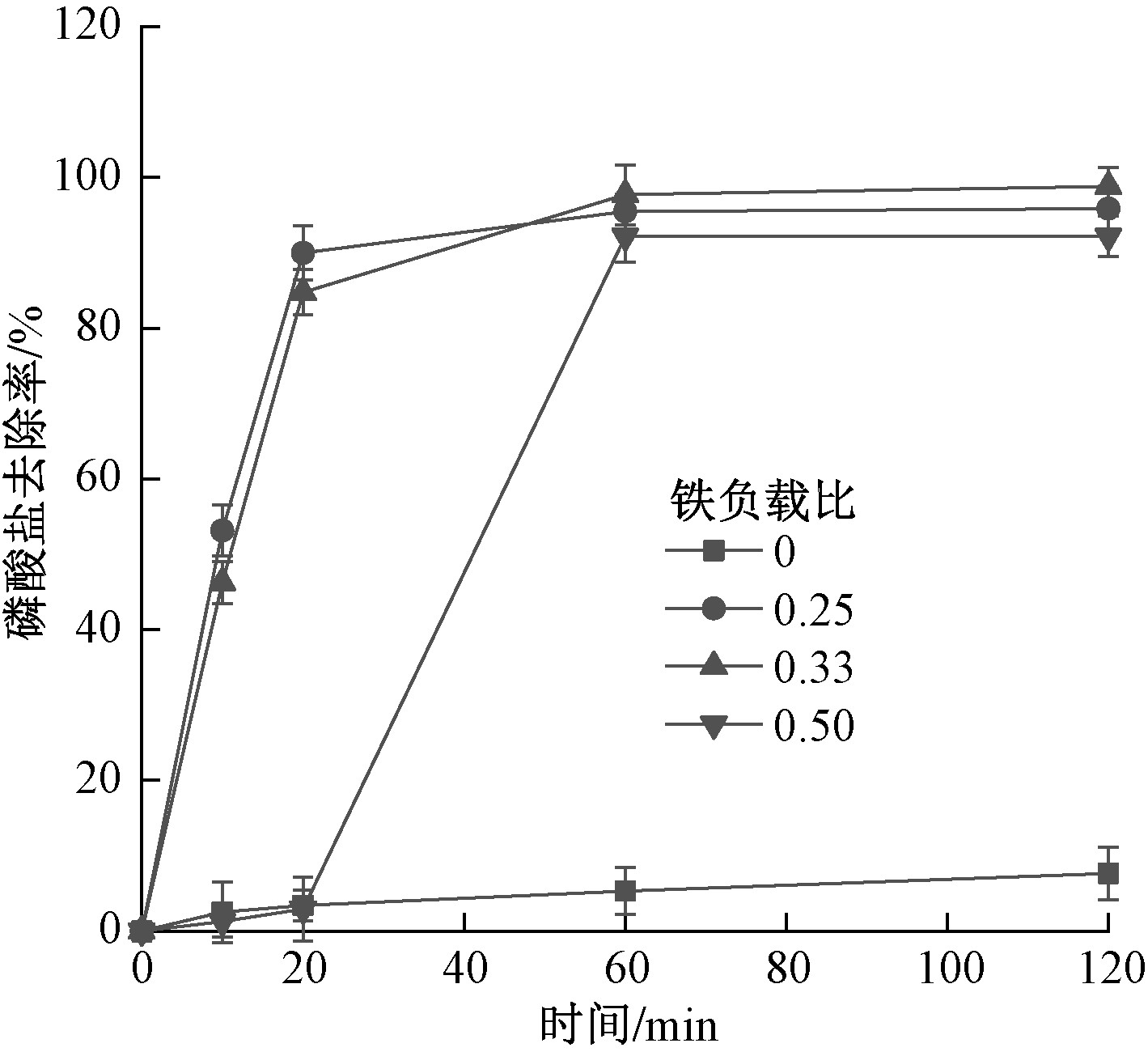
摘要: 以具有较大比表面积和良好吸附性能的天然杭锦土为载体制备杭锦土负载硫化零价铁(HJ@S-nZVI)。优化铁负载比、硫铁摩尔比(S/Fe)以及陈化时间等制备条件,利用扫描电子显微镜(SEM)、能量色散光谱(energy dispersive spectroscopy,EDS)、X射线光电子能谱(XPS)及比表面积(specific surface area,SSA)等手段对HJ@S-nZVI进行综合表征分析。考察投加量、初始pH以及共存离子等因素对HJ@S-nZVI去除磷酸盐效果的影响,并结合吸附等温线和吸附动力学研究其吸附性能和吸附机理。结果表明:HJ@S-nZVI的优化制备条件为铁负载比为0.25,S/Fe为0.01,陈化时间为10 d;SEM、EDS和元素分布图分析表明,硫化零价铁以球状颗粒形式成功负载于杭锦土表面,XPS表明HJ@S-nZVI表面铁的主要存在形态为FeS和FeOOH等;投加量、初始pH和SiO3 2−共存对HJ@S-nZVI去除磷酸盐的效果影响较大,而SO4 2−、CO3 2−和Cl−共存对磷酸盐的去除效果无明显竞争影响;HJ@S-nZVI对磷酸盐的吸附过程符合Freundlich等温模型(R2=0.992),不同初始浓度下,准二级动力学模型可较好地描述磷酸盐的去除过程(R2>0.995)。Abstract: Natural Hangjin clay with large specific surface area and excellent adsorption properties was used as a carrier to synthesize Hangjin clay-supported sulfidated zero-valent iron (HJ@S-nZVI). Preparation conditions such as the iron loading ratio, the molar ratio of sulfide iron (S/Fe) and the aging time were optimized. Scanning electron microscope (SEM), energy dispersive spectroscopy (EDS), X-ray photoelectron spectroscopy (XPS) and specific surface area (SSA) and other methods were utilized to comprehensively characterize and analyze the characteristics of HJ@S-nZVI. The effects of the dosage, initial pH, and co-existing ions on the phosphate removal efficiency were investigated, and the adsorption performance and adsorption mechanism of phosphate removal by HJ@S-nZVI were studied based on the adsorption isotherms and adsorption kinetics. The results showed that the optimized preparation conditions of HJ@S-nZVI were: iron loading ratio was 0.25, S/Fe was 0.01, and aging time was 10 d. SEM, EDS and element distribution map analysis manifested that sulfidated zero-valent iron was successfully loaded on the surface of Hangjin clay in the form of spherical particles. XPS analysis showed that the main existing forms of iron on the surface of HJ@S-nZVI were FeS and FeOOH. The dosage, initial pH and SiO3 2− had a greater influence on phosphate removal, while SO4 2−, CO3 2− and Cl− had no obvious competitive influence. The adsorption process of phosphate by HJ@S-nZVI conformed to Freundlich isotherm model (R2=0.992). Under different initial concentrations, the quasi-second-order kinetic model could better describe the phosphate removal process(R2>0.995).
-
Key words:
- sulfidated zero-valent iron /
- Hangjin clay /
- modification /
- adsorption /
- phosphate
-
表 1 HJ矿物组成与阳离子交换量[17]
Table 1. Mineral composition and cation exchange capacity of Hangjin clay
CEC/
(cmol/kg)矿物占比/% 烧失
量/%SiO2 Al2O3 CaO Fe2O3 MgO K2O Na2O TiO2 32.2 50.9 15.2 6.4 5.9 3.6 3.6 0.9 0.6 12.9 表 2 HJ及改性材料SSA、微孔体积及平均孔径
Table 2. Specific surface area, pore volume and average pore diameter of Hangjin clay and modified materials
样品 SSA/(m2/g) 微孔体积/(cm3/g) 平均孔径/nm HJ 66.59 0.1153 6.92 HJ@nZVI 42.07 0.0899 8.55 HJ@S-nZVI 47.34 0.0929 7.85 表 3 HJ@S-nZVI去除磷酸盐的等温吸附模型拟合参数
Table 3. Isotherm parameters for phosphate adsorption by HJ@S-nZVI
Langmuir等温吸附模型 Freundlich等温吸附模型 qm/(mg/g) KL/(L/mg) R2 KF n R2 32.86 2.168 0.9903 22.45 4.926 0.9920 表 4 HJ@S-nZVI去除磷酸盐的吸附动力学参数
Table 4. Kinetic parameters of phosphate adsorption by HJ@S-nZVI
磷酸盐浓度/
(mg/L)qe,exp/(mg/g) 准一级动力学模型 准二级动力学模型 k1/min−1 qe,cal/(mg/g) R2 k2/min−1 qe,cal/(mg/g) R2 5 9.82 0.055 9.44 0.939 0.012 0 9.89 0.999 10 19.07 0.052 17.78 0.937 0.003 7 19.27 0.999 15 25.25 0.035 22.93 0.909 0.001 6 25.61 0.999 20 29.15 0.012 28.15 0.949 0.000 7 30.08 0.998 25 29.81 0.009 28.54 0.889 0.000 6 30.68 0.995 注:qe,exp为试验得到的吸附平衡时的吸附容量;qe,cal为模型计算得到的吸附平衡时的吸附容量。 表 5 HJ@S-nZVI去除磷酸盐的Weber-Morris颗粒内扩散模型参数
Table 5. Parameters of the Weber-Morris diffusion model for phosphate removal by HJ@S-nZVI
磷酸盐
浓度/
(mg/L)第一阶段 第二阶段 第三阶段 kid1/〔mg/(g·min−0.5)〕 R2 kid2/〔mg/(g·min−0.5)〕 R2 kid3/〔mg/(g·min−0.5)〕 R2 5 1.39 0.971 0.45 0.935 0.003 8 0.723 10 2.53 0.962 0.74 0.958 0.060 0.840 15 2.89 0.999 0.92 0.969 0.18 0.804 20 2.03 0.965 1.41 0.971 0.30 0.823 25 2.40 0.949 1.16 0.940 0.39 0.870 -
[1] VENKITESHWARAN K, MCNAMARA P J, MAYER B K. Meta-analysis of non-reactive phosphorus in water, wastewater, and sludge, and strategies to convert it for enhanced phosphorus removal and recovery[J]. Science of the Total Environment,2018,644:661-674. doi: 10.1016/j.scitotenv.2018.06.369 [2] CHEN L, LI Y Z, SUN Y B, et al. La(OH)3 loaded magnetic mesoporous nanospheres with highly efficient phosphate removal properties and superior pH stability[J]. Chemical Engineering Journal,2019,360:342-348. doi: 10.1016/j.cej.2018.11.234 [3] REN J, LI N, WEI H, et al. Efficient removal of phosphorus from turbid water using chemical sedimentation by FeCl3 in conjunction with a starch-based flocculant[J]. Water Research,2020,170:115361. doi: 10.1016/j.watres.2019.115361 [4] 简志强, 周高婷, 龚斌, 等.微米零价铁去除磷酸盐效果与机理研究[J]. 环境工程技术学报,2021,11(5):927-934. doi: 10.12153/j.issn.1674-991X.20210027JIAN Z Q, ZHOU G T, GONG B, et al. Study on the efficacy of micron zero-valent iron on phosphate removal and its mechanism[J]. Journal of Environmental Engineering Technology,2021,11(5):927-934. doi: 10.12153/j.issn.1674-991X.20210027 [5] PENG X, XI B D, ZHAO Y, et al. Effect of arsenic on the formation and adsorption property of ferric hydroxide precipitates in ZVI treatment[J]. Environmental Science & Technology,2017,51(17):10100-10108. [6] WANG H Y, ZOU Z C, XIAO X L, et al. Reduction of highly concentrated phosphate from aqueous solution using pectin-nanoscale zerovalent iron (PNZVI)[J]. Water Science and Technology,2016,73(11):2689-2696. doi: 10.2166/wst.2016.106 [7] ZHANG Q, LIU H B, CHEN T H, et al. The synthesis of NZVI and its application to the removal of phosphate from aqueous solutions[J]. Water, Air, & Soil Pollution,2017,228(9):1-10. [8] MAHMOUD A S, MOSTAFA M K, NASR M. Regression model, artificial intelligence, and cost estimation for phosphate adsorption using encapsulated nanoscale zero-valent iron[J]. Separation Science and Technology,2019,54(1):13-26. doi: 10.1080/01496395.2018.1504799 [9] SONG S K, SU Y M, ADELEYE A S, et al. Optimal design and characterization of sulfide-modified nanoscale zerovalent iron for diclofenac removal[J]. Applied Catalysis B:Environmental,2017,201:211-220. doi: 10.1016/j.apcatb.2016.07.055 [10] BIN Q, LIN B, ZHU K, et al. Superior trichloroethylene removal from water by sulfide-modified nanoscale zero-valent iron/graphene aerogel composite[J]. Journal of Environmental Sciences,2020,88:90-102. doi: 10.1016/j.jes.2019.08.011 [11] LI Y J, ZHAO X G, YAN Y, et al. Enhanced sulfamethoxazole degradation by peroxymonosulfate activation with sulfide-modified microscale zero-valent iron (S-mFe0): performance, mechanisms, and the role of sulfur species[J]. Chemical Engineering Journal,2019,376:121302. doi: 10.1016/j.cej.2019.03.178 [12] RAYAROTH M P, PRASANTHKUMAR K P, KANG Y G, et al. Degradation of carbamazepine by singlet oxygen from sulfidized nanoscale zero-valent iron: citric acid system[J]. Chemical Engineering Journal,2020,382:122828. doi: 10.1016/j.cej.2019.122828 [13] WANG Y H, SHAO Q Q, HUANG S S, et al. High performance and simultaneous sequestration of Cr(Ⅵ) and Sb(Ⅲ) by sulfidated zerovalent iron[J]. Journal of Cleaner Production,2018,191:436-444. doi: 10.1016/j.jclepro.2018.04.217 [14] MA F F, ZHAO B W, DIAO J R, et al. Mechanism of phosphate removal from aqueous solutions by biochar supported nanoscale zero-valent iron[J]. RSC Advances,2020,10(64):39217-39225. doi: 10.1039/D0RA07391A [15] 蔡金水, 康得军, 杨天学, 等.铁改性杭锦土吸附剂对水中砷的去除研究[J]. 环境科学研究,2021,34(2):346-355.CAI J S, KANG D J, YANG T X, et al. Removal of arsenic from water by iron modified Hangjin clay adsorbent[J]. Research of Environmental Sciences,2021,34(2):346-355. [16] LI X G, ZHAO Y, XI B D, et al. Removal of nitrobenzene by immobilized nanoscale zero-valent iron: effect of clay support and efficiency optimization[J]. Applied Surface Science,2016,370:260-269. doi: 10.1016/j.apsusc.2016.01.141 [17] 李晓光. 杭锦土负载纳米零价铁的研制及其去除典型污染物效能与机理研究[R]. 北京: 北京师范大学, 2016. [18] 李春侠. 负载型硫化纳米零价铁去除水中As(Ⅲ)和Cu(Ⅱ)的研究[D]. 武汉: 武汉科技大学, 2019. [19] NAGOYA S, NAKAMICHI S, KAWASE Y. Mechanisms of phosphate removal from aqueous solution by zero-valent iron: a novel kinetic model for electrostatic adsorption, surface complexation and precipitation of phosphate under oxic conditions[J]. Separation and Purification Technology,2019,218:120-129. doi: 10.1016/j.seppur.2019.02.042 [20] LI X G, ZHAO Y, XI B D, et al. Decolorization of methyl orange by a new clay-supported nanoscale zero-valent iron: synergetic effect, efficiency optimization and mechanism[J]. Journal of Environmental Sciences,2017,52:8-17. doi: 10.1016/j.jes.2016.03.022 [21] KIM E J, KIM J H, AZAD A M, et al. Facile synthesis and characterization of Fe/FeS nanoparticles for environmental applications[J]. ACS Applied Materials & Interfaces,2011,3(5):1457-1462. [22] TURCIO-ORTEGA D, FAN D M, TRATNYEK P G, et al. Reactivity of Fe/FeS nanoparticles: electrolyte composition effects on corrosion electrochemistry[J]. Environmental Science & Technology,2012,46(22):12484-12492. [23] XU B D, LI D C, QIAN T T, et al. Boosting the activity and environmental stability of nanoscale zero-valent iron by montmorillonite supporting and sulfidation treatment[J]. Chemical Engineering Journal,2020,387:124063. doi: 10.1016/j.cej.2020.124063 [24] 刘文芳, 赵颖, 蔡亚君, 等.杭锦土负载纳米零价铁对水中甲基橙的脱色研究[J]. 工业水处理,2015,35(12):34-39. doi: 10.11894/1005-829x.2015.35(12).034LIU W F, ZHAO Y, CAI Y J, et al. Study on the decolorization of methyl orange in aqueous solution using Hangjin clay-supported nanoscale zero-valent iron[J]. Industrial Water Treatment,2015,35(12):34-39. doi: 10.11894/1005-829x.2015.35(12).034 [25] 王亚浩, 邵欠欠, 张练, 等.膨润土负载硫化纳米零价铁去除对硝基苯酚的研究[J]. 环境污染与防治,2019,41(3):329-333.WANG Y H, SHAO Q Q, ZHANG L, et al. The removal of p-nitrophenol by sulfidated nano-zero valent iron supported by bentonite[J]. Environmental Pollution & Control,2019,41(3):329-333. [26] PANG Z H, YAN M Y, JIA X S, et al. Debromination of decabromodiphenyl ether by organo-montmorillonite-supported nanoscale zero-valent iron: preparation, characterization and influence factors[J]. Journal of Environmental Sciences,2014,26(2):483-491. doi: 10.1016/S1001-0742(13)60419-2 [27] LIU W, AI Z H, CAO M H, et al. Ferrous ions promoted aerobic simazine degradation with Fe@Fe2O3 core-shell nanowires[J]. Applied Catalysis B:Environmental,2014,150/151:1-11. doi: 10.1016/j.apcatb.2013.11.034 [28] DONG J, ZHAO Y S, ZHAO R, et al. Effects of pH and particle size on kinetics of nitrobenzene reduction by zero-valent iron[J]. Journal of Environmental Sciences,2010,22(11):1741-1747. doi: 10.1016/S1001-0742(09)60314-4 [29] BOPARAI H K, JOSEPH M, O’CARROLL D M. Kinetics and thermodynamics of cadmium ion removal by adsorption onto nano zerovalent iron particles[J]. Journal of Hazardous Materials,2011,186(1):458-465. ◇ doi: 10.1016/j.jhazmat.2010.11.029 -





 下载:
下载: